We stock a comprehensive range of gauge sizes strictly adhering to ISO 6009 colour coding standards.
NOTE: We've listed the most common types below, however you can view our product catalogue to see all gauges we are able to supply.




Certified & Approved Products
M4 Medical Supplies & Logistics offers healthcare providers with ISO-compliant hypodermic equipment and sterile consumables. Fast, certified, and ready for bulk deployment to anywhere in the world.



Every product meets rigorous safety standards and is compliant with globally recognised compliance standards, ensuring sterility and patient safety from the warehouse to the ward.
We understand that medical inventory is time-critical. Our logistics network ensures reduced lead times for urgent procurement, whether by land, air or sea.
Built for clinics, hospitals, and pharmacies. We specialise in high-volume orders with consistent stock availability.

We stock a comprehensive range of gauge sizes strictly adhering to ISO 6009 colour coding standards.
NOTE: We've listed the most common types below, however you can view our product catalogue to see all gauges we are able to supply.
Drawing & Mixing
In Stock
Standard Injection
In Stock
Pediatric Use
In Stock
Subcutaneous
In Stock
Insulin/Fine
In Stock
Browse an overview of all products we are able to procure and ship in bulk, on-demand.
To place an order efficiently, please contact us directly via our WhatsApp line.




Engagements with M4 Medical Supplies and Logistics begin through direct coordination with authorized representatives.
To initiate a discussion, prospective clients should be prepared to outline:
Once initial information is received, M4 conducts an internal review to assess feasibility, timelines, and logistical requirements. From there, qualified engagements proceed through formal order structuring, documentation review, and coordinated fulfillment planning.
M4 is structured to support governmental, sovereign, and humanitarian clients, and all engagements are handled with professionalism, discretion, and clarity from initial inquiry through delivery.
Sample requests may be supported for qualified institutional, governmental, or procurement-led evaluations, subject to product availability and review.
Sample quantities, product types, and delivery arrangements are assessed on a case-by-case basis to support procurement decisions while maintaining alignment with large-scale supply operations.
All sample requests are coordinated through formal engagement with authorized representatives.
To maintain a secure and uncompromised medical chain of custody, M4 Medical Supplies and Logistics does not accept returns of sterile goods once they have left controlled handling, except in cases of verified manufacturing defect.
This policy ensures that all products delivered are factory-sealed, uncompromised, and compliant with sterility and safety requirements.
Return and remediation procedures, where applicable, are addressed through formal quality review.
M4 Medical Supplies and Logistics primarily transacts in United States Dollars (USD).
Alternative currencies may be considered on a case-by-case basis, depending on transaction structure, jurisdiction, and banking requirements. Any approved currency arrangements are confirmed during the order review and contracting process.
Yes. M4 Medical Supplies and Logistics provides standard export and shipment documentation required to support customs clearance.
This may include commercial invoices, packing lists, certificates of origin, sterility certifications, and other documentation as required based on destination and regulatory considerations.
Documentation requirements are reviewed and confirmed during logistics planning to support efficient clearance in the destination country.
Yes. M4 supplies a range of safety-engineered medical devices, including retractable and safety-shielded designs intended to reduce the risk of needlestick injuries.
These products are manufactured in accordance with applicable international safety and occupational health standards and are suitable for use in institutional, governmental, and humanitarian healthcare environments.
Availability is subject to product category and order specifications.
Sterile products supplied through M4 Medical Supplies and Logistics typically carry a shelf life of 3 to 5 years, depending on product type and manufacturing specifications.
This extended shelf life supports long-term storage, emergency preparedness, and deployment to remote or underserved regions, provided products are stored and handled in accordance with recommended conditions and packaging remains intact.
Shelf-life details are confirmed at the time of order fulfillment.
Yes. Our consumable products are designed to be compatible with standard medical equipment used in regulated healthcare environments.
This includes universal connection systems manufactured in accordance with applicable ISO standards, ensuring consistent interoperability across hospitals, clinics, and field operations globally.
Product specifications and compatibility details are confirmed during order evaluation.
Yes. Medical devices supplied through M4 Medical Supplies and Logistics are CE marked, indicating conformity with applicable European Union health, safety, and performance requirements.
CE certification is widely recognized across international markets and supports regulatory acceptance in multiple regions worldwide. Relevant compliance documentation is provided as part of the order review and fulfillment process.
Yes. All products supplied through M4 Medical Supplies and Logistics are manufactured in accordance with recognized international medical standards and regulatory requirements.
This includes compliance with applicable ISO standards for sterile medical devices and globally accepted product classification and identification systems, ensuring consistency and recognition across international healthcare environments.
Product certifications and compliance documentation are provided as required during order review and fulfillment.
For international shipments, all medical products are packaged in export-grade, reinforced outer packaging designed to withstand extended transit conditions, including environmental exposure and handling variability.
M4 adheres to Good Distribution Practice (GDP) standards to preserve product integrity, documentation consistency, and chain-of-custody control across international borders.
Packaging specifications are aligned with regulatory, customs, and destination-country requirements.
Yes. M4 Medical Supplies and Logistics supports delivery to remote, underserved, and hard-to-reach locations as part of government-led, humanitarian, and emergency response initiatives.
We coordinate specialized logistics solutions to support delivery beyond major population centers, including regions affected by conflict, infrastructure limitations, or geographic constraints.
Delivery planning is conducted in coordination with authorized representatives to ensure feasibility, security, and compliance with destination-specific requirements.
Yes. M4 Medical Supplies and Logistics supports structured, recurring supply arrangements for high-volume medical consumables as part of long-term national, institutional, and humanitarian supply programs.
For qualified clients, we offer standing order frameworks designed to support consistent supply planning, reduce the risk of shortages, and streamline fulfillment over time.
These arrangements are particularly suited for international and government-led programs, where coordinated scheduling, documentation consistency, and reliable delivery are critical.
Recurring order structures are established based on volume, product category, regulatory considerations, and destination requirements, and are confirmed through formal agreement.
We operate through a centralized global distribution model that allows us to support clients worldwide while maintaining flexibility for varying order requirements.
Delivery timelines depend on several factors, including product type, order volume, and destination. Typical lead times are as follows:
For bulk purchases or customized supply requests, timelines may vary to ensure proper handling, quality assurance, and coordinated logistics. Our team works closely with each client to provide clear delivery expectations at the time of order confirmation.
For the most accurate estimates, we recommend consulting with our logistics team based on your specific order needs.
Delivery timelines provided are estimates and may be subject to change based on order complexity, regulatory requirements, carrier availability, and destination-specific logistics.
Yes. M4 Medical Supplies and Logistics is structured to support large-scale national, governmental, and humanitarian medical supply programs.
Our operations are optimized for high-volume bulk procurement, typically 10 million units or more, supplied in full cartons and palletized freight to ensure consistency, quality control, and logistical efficiency.
Requests below standard bulk volumes are not aligned with our primary operating model and are considered only under exceptional circumstances, such as urgent humanitarian needs or government-directed initiatives, and are subject to internal review and approval.
Minimum order quantities, packaging standards, and fulfillment terms are confirmed during formal order evaluation and may vary based on urgency, product category, and destination requirements.
M4 Medical Supplies and Logistics serves governmental, sovereign, and institutional clients requiring large-scale medical supply solutions.
Our primary clients include:
Our operations are designed to support high-volume, mission-critical medical supply programs, including urgent response scenarios, national procurement efforts, and large-scale humanitarian needs.
We work closely with authorized representatives to ensure secure coordination, clear communication, and dependable fulfillment aligned with governmental and institutional requirements.
Louisville, KY


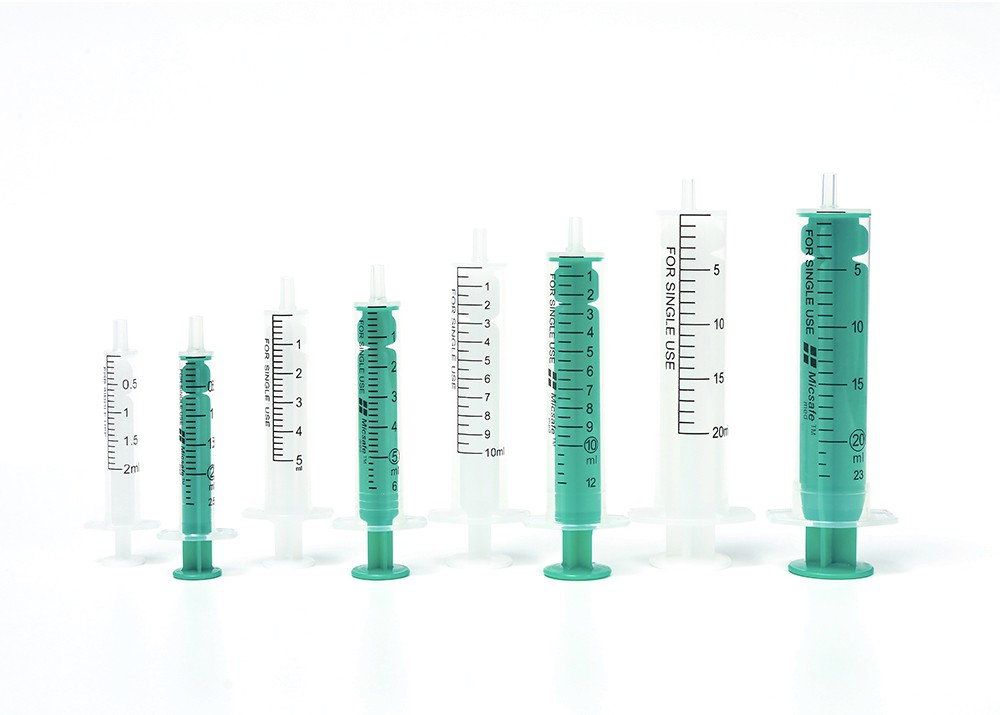


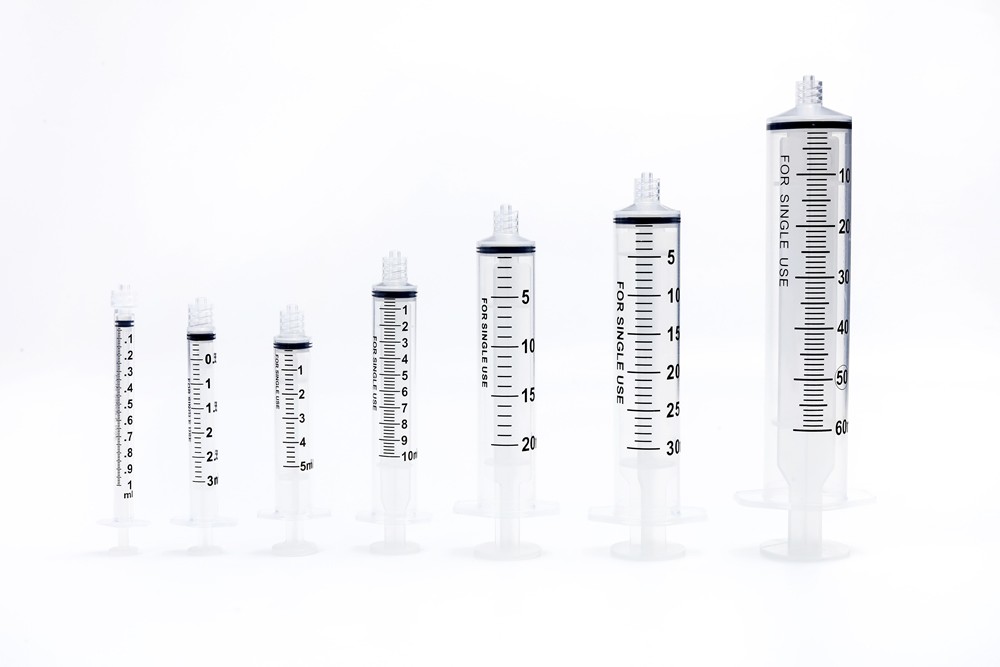
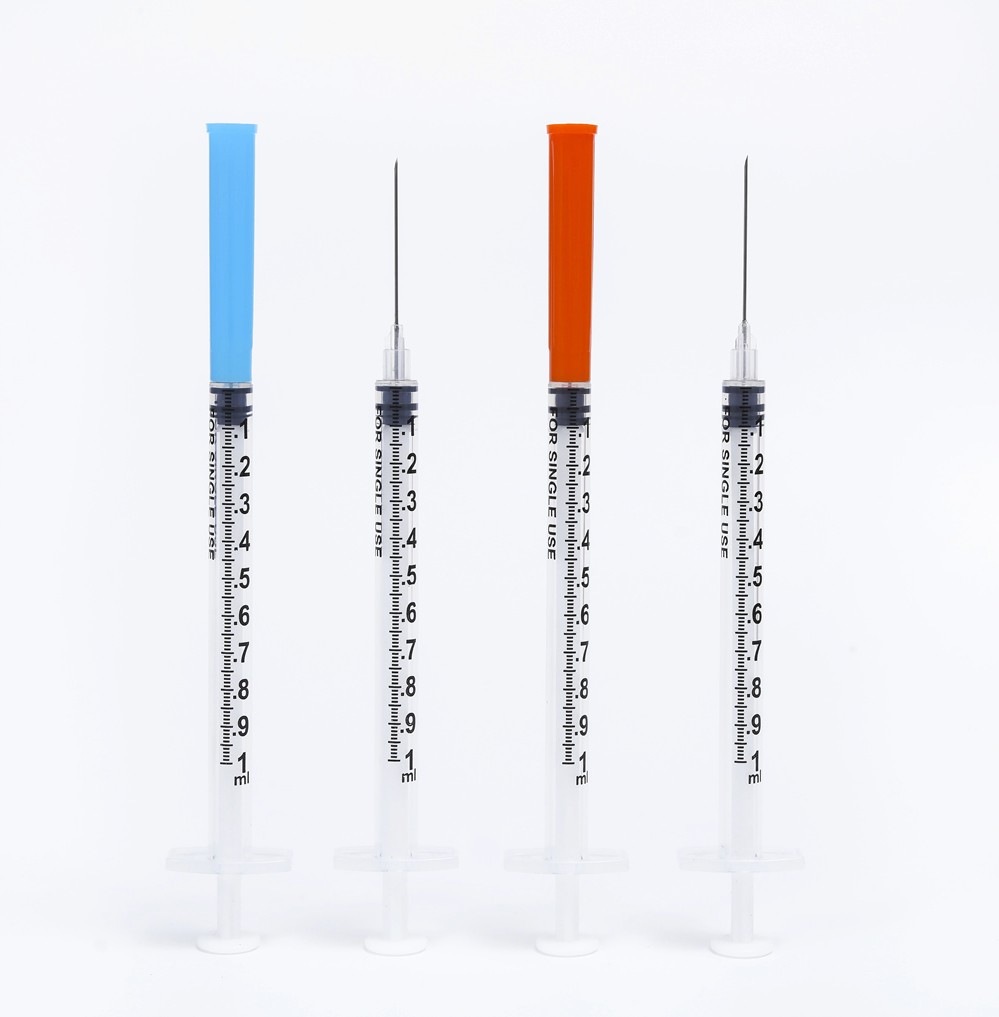







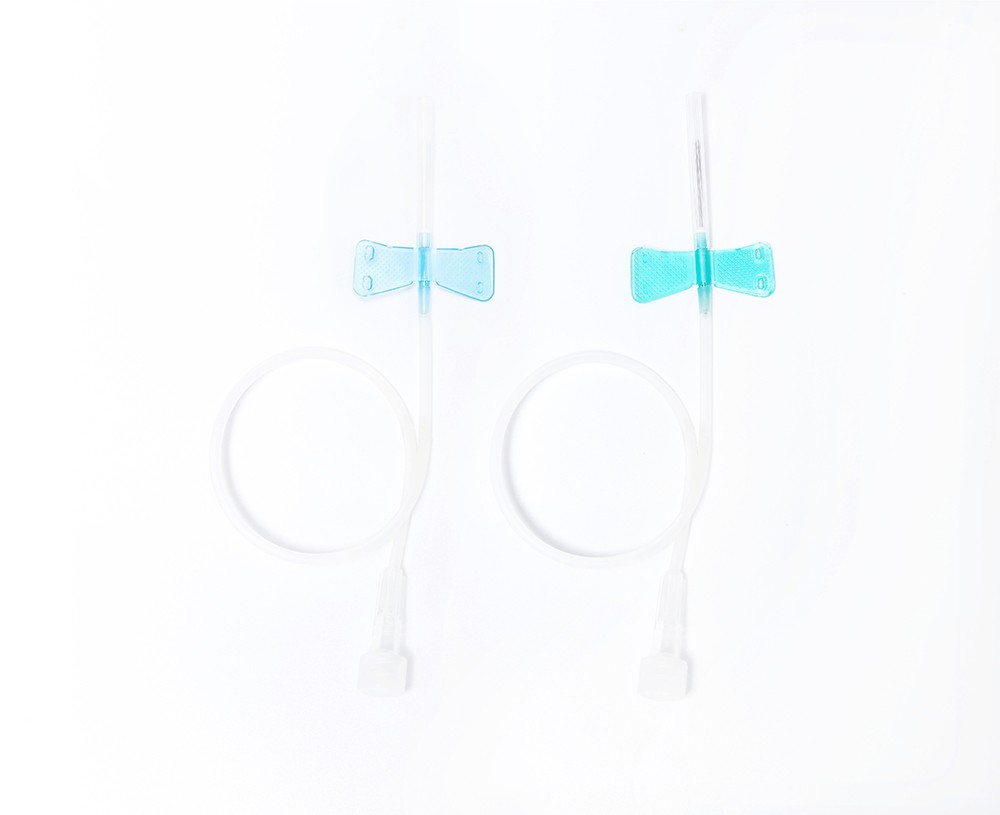
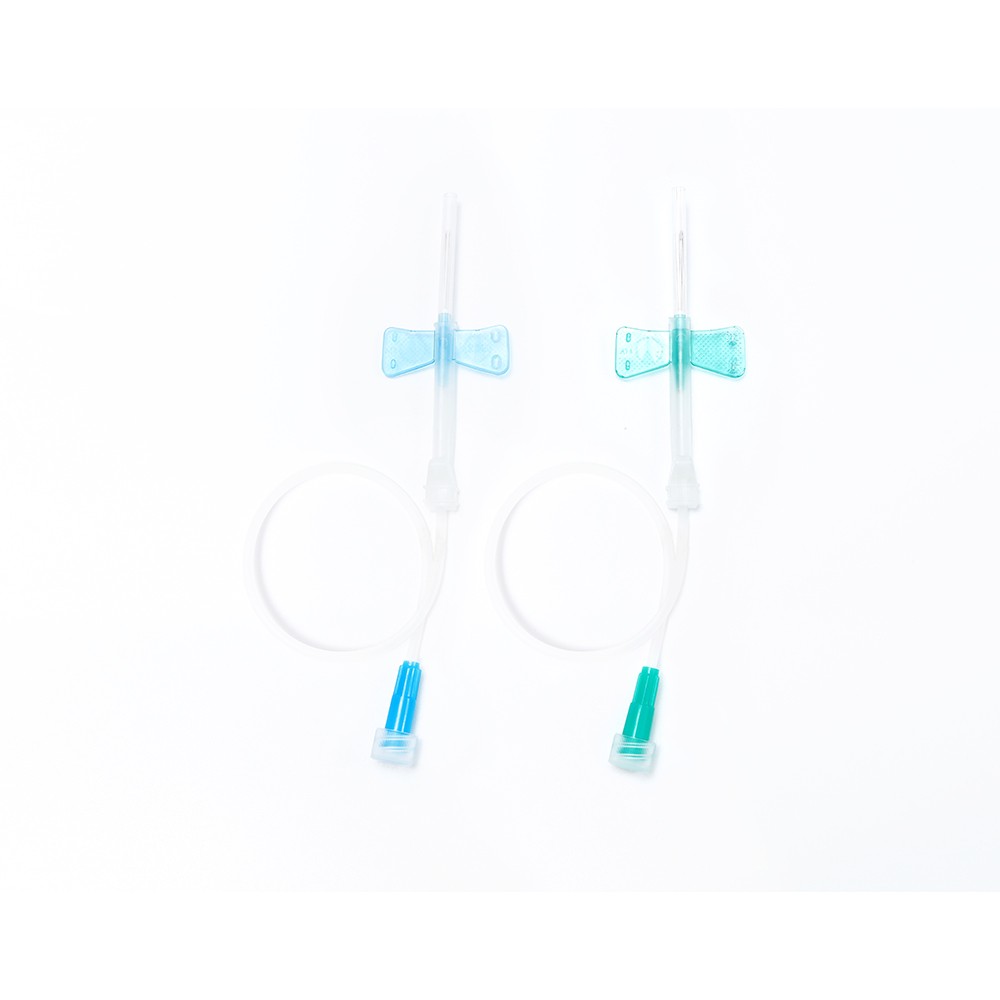
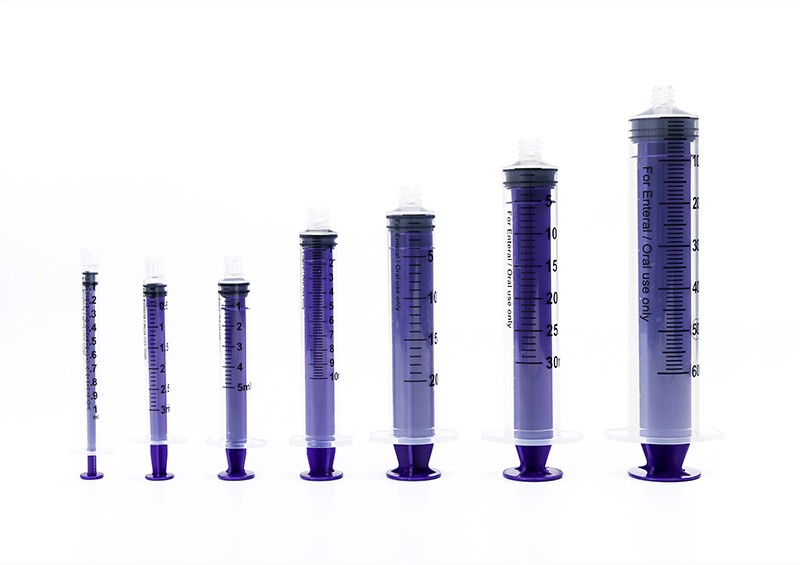



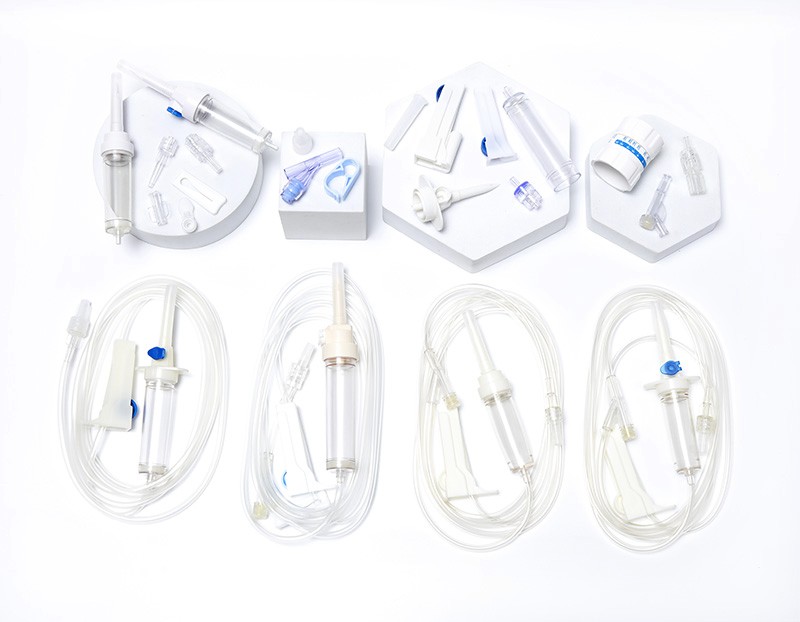
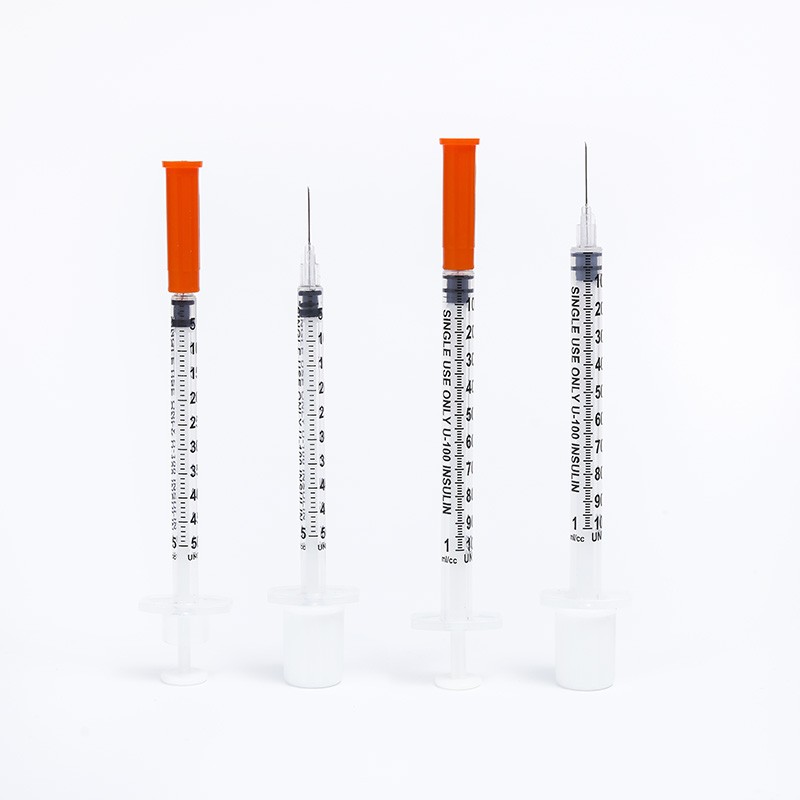

O-Ring Oral Syringe 20ml
OR20MT
Spec length
20ml
Spec length
-
Spec Tip
Oral Tip
Sub Group
Standard
Spec Gauge
Units per box
50/box
O-Ring Oral Syringe 60ml
OR60MT
Spec length
60ml
Spec length
-
Spec Tip
Oral Tip
Sub Group
Standard
Spec Gauge
Units per box
30/box
Syringe 5-6ml Centric (3-Part Luer)
SY20C3L
Spec length
5-6ml
Spec length
-
Spec Tip
Centric
Sub Group
Standard
Spec Gauge
Units per box
100/box
Disposable Oral Syringe 5ml
OR05ST
Spec length
5ml
Spec length
-
Spec Tip
Oral Tip
Sub Group
Standard
Spec Gauge
Units per box
100/box
Syringe 20ml Eccentric (2-Part)
SY20E2S
Spec length
20ml
Spec length
-
Spec Tip
Eccentric
Sub Group
Standard
Spec Gauge
Units per box
100/box
Syringe 10ml Eccentric (2-Part)
SY05E2S
Spec length
10ml
Spec length
-
Spec Tip
Eccentric
Sub Group
Standard
Spec Gauge
Units per box
100/box
Syringe 3ml Centric (3-Part Luer)
SY03C3L
Spec length
3ml
Spec length
-
Spec Tip
Centric
Sub Group
Standard
Spec Gauge
Units per box
100/box
Syringe 1ml Centric (3-Part)
SY01C3L
Spec length
1ml
Spec length
-
Spec Tip
Centric
Sub Group
Standard
Spec Gauge
Units per box
100/box
Syringe 30-35ml Eccentric (3-Part)
SY30E3S
Spec length
30-35ml
Spec length
-
Spec Tip
Eccentric
Sub Group
Standard
Spec Gauge
Units per box
40/box
Syringe 30-35ml Centric (3-Part)
SY30C3S
Spec length
30-35ml
Spec length
-
Spec Tip
Centric
Sub Group
Standard
Spec Gauge
Units per box
40/box
Syringe 10-12ml Eccentric (3-Part)
SY10E3S
Spec length
10-12ml
Spec length
-
Spec Tip
Eccentric
Sub Group
Standard
Spec Gauge
Units per box
100/box
Syringe 10-12ml Centric (3-Part)
SY10C3S
Spec length
10-12ml
Spec length
-
Spec Tip
Centric
Sub Group
Standard
Spec Gauge
Units per box
100/box
Syringe 2-3ml Centric (3-Part)
SY03C3S
Spec length
2-3ml
Spec length
-
Spec Tip
Centric
Sub Group
Standard
Spec Gauge
Units per box
100/box
Prefilled Syringe 10-12ml
SY10P
Spec length
10-12ml
Spec length
-
Spec Tip
Centric
Sub Group
Standard
Spec Gauge
Units per box
100/box
Syringe 1ml Centric (O-Ring)
SY01C6L
Spec length
1ml
Spec length
-
Spec Tip
Centric
Sub Group
Standard
Spec Gauge
Units per box
100/box
Syringe 2-2.5ml Centric (O-Ring)
SY02C6S
Spec length
2-2.5ml
Spec length
-
Spec Tip
Centric
Sub Group
Standard
Spec Gauge
Units per box
100/box
Irrigation Syringe 100ml
IY100RT
Spec length
100ml
Spec length
-
Spec Tip
Ring-top
Sub Group
Standard
Spec Gauge
Units per box
50/box
Irrigation Syringe 60ml
IY60RT
Spec length
60ml
Spec length
-
Spec Tip
Ring-top
Sub Group
Standard
Spec Gauge
Units per box
50/box
O-Ring Oral Syringe 30ml
OR30MT
Spec length
30ml
Spec length
-
Spec Tip
Oral Tip
Sub Group
Standard
Spec Gauge
Units per box
50/box
O-Ring Oral Syringe 10ml
OR10MT
Spec length
10ml
Spec length
-
Spec Tip
Oral Tip
Sub Group
Standard
Spec Gauge
Units per box
100/box
O-Ring Oral Syringe 5ml
OR05MT
Spec length
5ml
Spec length
-
Spec Tip
Oral Tip
Sub Group
Standard
Spec Gauge
Units per box
100/box
O-Ring Oral Syringe 3ml
OR03MT
Spec length
3ml
Spec length
-
Spec Tip
Oral Tip
Sub Group
Standard
Spec Gauge
Units per box
100/box
O-Ring Oral Syringe 1ml
OR01MT
Spec length
1ml
Spec length
-
Spec Tip
Oral Tip
Sub Group
Standard
Spec Gauge
Units per box
100/box
Home Enteral Syringe 60ml
ER60MT
Spec length
60ml
Spec length
-
Spec Tip
Enteral Enfit
Sub Group
Home Use
Spec Gauge
Units per box
30/box
Home Enteral Syringe 30ml
ER30MT
Spec length
30ml
Spec length
-
Spec Tip
Enteral Enfit
Sub Group
Home Use
Spec Gauge
Units per box
50/box
Home Enteral Syringe 20ml
ER20MT
Spec length
20ml
Spec length
-
Spec Tip
Enteral Enfit
Sub Group
Home Use
Spec Gauge
Units per box
50/box
Home Enteral Syringe 10ml
ER10MT
Spec length
10ml
Spec length
-
Spec Tip
Enteral Enfit
Sub Group
Home Use
Spec Gauge
Units per box
100/box
Home Enteral Syringe 5ml
ER05MT
Spec length
5ml
Spec length
-
Spec Tip
Enteral Enfit
Sub Group
Home Use
Spec Gauge
Units per box
100/box
Home Enteral Syringe 3ml
ER03MT
Spec length
3ml
Spec length
-
Spec Tip
Enteral Enfit
Sub Group
Home Use
Spec Gauge
Units per box
100/box
Home Enteral Syringe 1ml
ER01MT
Spec length
1ml
Spec length
-
Spec Tip
Enteral Enfit
Sub Group
Home Use
Spec Gauge
Units per box
100/box
Disposable Oral Syringe 60ml
OR60ST
Spec length
60ml
Spec length
-
Spec Tip
Oral Tip
Sub Group
Standard
Spec Gauge
Units per box
30/box
Disposable Oral Syringe 30ml
OR30ST
Spec length
30ml
Spec length
-
Spec Tip
Oral Tip
Sub Group
Standard
Spec Gauge
Units per box
50/box
Disposable Oral Syringe 20ml
OR20ST
Spec length
20ml
Spec length
-
Spec Tip
Oral Tip
Sub Group
Standard
Spec Gauge
Units per box
50/box
Disposable Oral Syringe 10ml
OR10ST
Spec length
10ml
Spec length
-
Spec Tip
Oral Tip
Sub Group
Standard
Spec Gauge
Units per box
100/box
Disposable Oral Syringe 3ml
OR03ST
Spec length
3ml
Spec length
-
Spec Tip
Oral Tip
Sub Group
Standard
Spec Gauge
Units per box
100/box
Enteral Syringe 30ml (Clinical)
ER30ST
Spec length
30ml
Spec length
-
Spec Tip
Enteral Enfit
Sub Group
Standard
Spec Gauge
Units per box
50/box
Disposable Oral Syringe 1ml
OR01ST
Spec length
1ml
Spec length
-
Spec Tip
Oral Tip
Sub Group
Standard
Spec Gauge
Units per box
100/box
Enteral Syringe 60ml (Clinical)
ER50ST
Spec length
60ml
Spec length
-
Spec Tip
Enteral Enfit
Sub Group
Standard
Spec Gauge
Units per box
30/box
Enteral Syringe 20ml (Clinical)
ER20ST
Spec length
20ml
Spec length
-
Spec Tip
Enteral Enfit
Sub Group
Standard
Spec Gauge
Units per box
50/box
Enteral Syringe 10ml (Clinical)
ER10ST
Spec length
10ml
Spec length
-
Spec Tip
Enteral Enfit
Sub Group
Standard
Spec Gauge
Units per box
100/box
Enteral Syringe 5ml (Clinical)
ER05ST
Spec length
5ml
Spec length
-
Spec Tip
Enteral Enfit
Sub Group
Standard
Spec Gauge
Units per box
100/box
Enteral Syringe 3ml (Clinical)
ER03ST
Spec length
3ml
Spec length
-
Spec Tip
Enteral Enfit
Sub Group
Standard
Spec Gauge
Units per box
100/box
Enteral Syringe 1ml (Clinical)
ER01ST
Spec length
1ml
Spec length
-
Spec Tip
Enteral Enfit
Sub Group
Standard
Spec Gauge
Units per box
100/box
Veterinary Infusion Set B
IVET-CP02P
Spec length
-
Spec length
-
Spec Tip
-
Sub Group
Standard
Spec Gauge
Units per box
Vet Type B
Veterinary Infusion Set A
IVET-CP01P
Spec length
-
Spec length
-
Spec Tip
-
Sub Group
Standard
Spec Gauge
Units per box
Vet Type A
Transfusion Set B
TS-CP02P
Spec length
-
Spec length
-
Spec Tip
-
Sub Group
Standard
Spec Gauge
Units per box
Type B
Transfusion Set A
TS-CP01P
Spec length
-
Spec length
-
Spec Tip
-
Sub Group
Standard
Spec Gauge
Units per box
Type A
Blood Tube Plain
-
Spec length
-
Spec length
N/A
Spec Tip
Sub Group
Standard
Spec Gauge
Red Cap
Units per box
1800/ctn
Infusion Set Flow Regulator
IVAD-CP07P
Spec length
-
Spec length
-
Spec Tip
-
Sub Group
Standard
Spec Gauge
Units per box
Flow Reg
Infusion Set Flow Reg + Filter
IVAD-CP08P
Spec length
-
Spec length
-
Spec Tip
-
Sub Group
Standard
Spec Gauge
Units per box
Flow Reg+Filter
Infusion Set Vented + Filter
IVAD-CP04P
Spec length
-
Spec length
-
Spec Tip
-
Sub Group
Standard
Spec Gauge
Units per box
Vented+Filter
Infusion Set Vented
IVAD-CP03P
Spec length
-
Spec length
-
Spec Tip
-
Sub Group
Standard
Spec Gauge
Units per box
Vented
Blood Tube SST
-
Spec length
-
Spec length
N/A
Spec Tip
Sub Group
Standard
Spec Gauge
Orange Cap
Units per box
1800/ctn
Blood Tube EDTA
-
Spec length
-
Spec length
N/A
Spec Tip
-
Sub Group
Standard
Spec Gauge
Purple Cap
Units per box
1800/ctn
Blood Tube ESR
-
Spec length
-
Spec length
N/A
Spec Tip
Sub Group
Standard
Spec Gauge
Black Cap
Units per box
1800/ctn
Blood Tube Glucose
-
Spec length
-
Spec length
N/A
Spec Tip
Sub Group
Standard
Spec Gauge
Grey Cap
Units per box
1800/ctn
Blood Tube Heparin
-
Spec length
-
Spec length
N/A
Spec Tip
Sub Group
Standard
Spec Gauge
Green Cap
Units per box
1800/ctn
Blood Tube Sodium Citrate
-
Spec length
-
Spec length
N/A
Spec Tip
Sub Group
Standard
Spec Gauge
Blue Cap
Units per box
1800/ctn
Insulin Pen Needle 33G 4mm
IPN3304
Spec length
-
Spec length
4mm
Spec Tip
-
Sub Group
Standard
Spec Gauge
33G
Units per box
100/box
Insulin Pen Needle 32G 4mm
IPN3204
Spec length
-
Spec length
4mm
Spec Tip
-
Sub Group
Standard
Spec Gauge
32G
Units per box
100/box
Insulin Pen Needle 31G 4mm
IPN3104
Spec length
-
Spec length
4mm
Spec Tip
-
Sub Group
Standard
Spec Gauge
31G
Units per box
100/box
Insulin Pen Needle 31G 6mm
IPN3106
Spec length
-
Spec length
6mm
Spec Tip
-
Sub Group
Standard
Spec Gauge
31G
Units per box
100/box
Insulin Pen Needle 31G 8mm
IPN3108
Spec length
-
Spec length
8mm
Spec Tip
-
Sub Group
Standard
Spec Gauge
31G
Units per box
100/box
Insulin Pen Needle 30G 8mm
IPN3008
Spec length
-
Spec length
8mm
Spec Tip
-
Sub Group
Standard
Spec Gauge
30G
Units per box
100/box
Safety Needle 30G 5/8"
DN3058S
Spec length
-
Spec length
5/8"
Spec Tip
Luer
Sub Group
Safety
Spec Gauge
30G
Units per box
100/box
Safety Needle 30G 1/2"
DN3012S
Spec length
-
Spec length
1/2"
Spec Tip
Luer
Sub Group
Safety
Spec Gauge
30G
Units per box
100/box
Disposable Needle 30G 1/2"
DN3012
Spec length
-
Spec length
1/2"
Spec Tip
Luer
Sub Group
Standard
Spec Gauge
30G
Units per box
100/box
Disposable Needle 30G 3/4"
DN3034
Spec length
-
Spec length
3/4"
Spec Tip
Luer
Sub Group
Standard
Spec Gauge
30G
Units per box
100/box
Insulin Syringe 1ml
ISY01F
Spec length
1ml
Spec length
-
Spec Tip
Fixed
Sub Group
Standard
Spec Gauge
27-32G
Units per box
100/box
Insulin Pen Needle 29G 13mm
IPN2913
Spec length
-
Spec length
13mm
Spec Tip
-
Sub Group
Standard
Spec Gauge
29G
Units per box
100/box
Disposable Needle 27G 1 1/2"
DN27112
Spec length
-
Spec length
1 1/2"
Spec Tip
Luer
Sub Group
Standard
Spec Gauge
27G
Units per box
100/box
Disposable Needle 27G 1/2"
DN2712
Spec length
-
Spec length
1/2"
Spec Tip
Luer
Sub Group
Standard
Spec Gauge
27G
Units per box
100/box
Disposable Needle 27G 3/4"
DN2734
Spec length
-
Spec length
3/4"
Spec Tip
Luer
Sub Group
Standard
Spec Gauge
27G
Units per box
100/box
Insulin Syringe 0.5ml
ISY005F
Spec length
0.5ml
Spec length
-
Spec Tip
Fixed
Sub Group
Standard
Spec Gauge
27-32G
Units per box
100/box
Insulin Syringe 0.3ml
ISY003F
Spec length
0.3ml
Spec length
-
Spec Tip
Fixed
Sub Group
Standard
Spec Gauge
27-32G
Units per box
100/box
Safety Insulin Syringe 1ml
ISY01FS
Spec length
1ml
Spec length
-
Spec Tip
Fixed
Sub Group
Standard
Spec Gauge
27-32G
Units per box
100/box
Safety Insulin Syringe 0.5ml
ISY005FS
Spec length
0.5ml
Spec length
-
Spec Tip
Fixed
Sub Group
Standard
Spec Gauge
27-32G
Units per box
100/box
Safety Insulin Syringe 0.3ml
ISY003FS
Spec length
0.3ml
Spec length
-
Spec Tip
Fixed
Sub Group
Standard
Spec Gauge
27-32G
Units per box
100/box
Safety Needle 26G 1"
DN2601S
Spec length
-
Spec length
1"
Spec Tip
Luer
Sub Group
Safety
Spec Gauge
26G
Units per box
100/box
Safety Needle 26G 1/2"
DN2612S
Spec length
-
Spec length
1/2"
Spec Tip
Luer
Sub Group
Safety
Spec Gauge
26G
Units per box
100/box
Disposable Needle 26G 5/8"
DN2658
Spec length
-
Spec length
5/8"
Spec Tip
Luer
Sub Group
Standard
Spec Gauge
26G
Units per box
100/box
Disposable Needle 26G 3/8"
DN2638
Spec length
-
Spec length
3/8"
Spec Tip
Luer
Sub Group
Standard
Spec Gauge
26G
Units per box
100/box
Fixed Needle Syringe 1ml 25G
SY01F2501
Spec length
1ml
Spec length
-
Spec Tip
Fixed
Sub Group
Standard
Spec Gauge
25G
Units per box
100/box
Disposable Needle 26G 1/2"
DN2612
Spec length
-
Spec length
1/2"
Spec Tip
Luer
Sub Group
Standard
Spec Gauge
26G
Units per box
100/box
Safety Butterfly Needle 25G 3/4"
BTF2534S
Spec length
-
Spec length
3/4"
Spec Tip
Luer
Sub Group
Standard
Spec Gauge
25G
Units per box
100/box
Safety Needle 23G 1"
DN2301S
Spec length
-
Spec length
1"
Spec Tip
Luer
Sub Group
Safety
Spec Gauge
23G
Units per box
100/box
Butterfly Needle 25G 3/4"
BTF2534
Spec length
-
Spec length
3/4"
Spec Tip
Luer
Sub Group
Standard
Spec Gauge
25G
Units per box
100/box
BC Butterfly 25G 3/4"
BC2534B
Spec length
-
Spec length
3/4"
Spec Tip
Luer
Sub Group
Standard
Spec Gauge
25G
Units per box
50/box
Safety BC Butterfly 25G 3/4"
BC2534BS
Spec length
-
Spec length
3/4"
Spec Tip
Luer
Sub Group
Standard
Spec Gauge
25G
Units per box
50/box
Safety Butterfly Needle 23G 3/4"
BTF2334S
Spec length
-
Spec length
3/4"
Spec Tip
Luer
Sub Group
Standard
Spec Gauge
23G
Units per box
100/box
Safety Needle 23G 1/2"
DN2312S
Spec length
-
Spec length
1/2"
Spec Tip
Luer
Sub Group
Safety
Spec Gauge
23G
Units per box
100/box
Disposable Needle 23G 1 1/4"
DN23114
Spec length
-
Spec length
1 1/4"
Spec Tip
Luer
Sub Group
Standard
Spec Gauge
23G
Units per box
100/box
Disposable Needle 23G 1"
DN2301
Spec length
-
Spec length
1"
Spec Tip
Luer
Sub Group
Standard
Spec Gauge
23G
Units per box
100/box
Disposable Needle 23G 3/4"
DN2334
Spec length
-
Spec length
3/4"
Spec Tip
Luer
Sub Group
Standard
Spec Gauge
23G
Units per box
100/box
Butterfly Needle 23G 3/4"
BTF2334
Spec length
-
Spec length
3/4"
Spec Tip
Luer
Sub Group
Standard
Spec Gauge
23G
Units per box
100/box
Safety BC Butterfly 23G 3/4"
BC2334BS
Spec length
-
Spec length
3/4"
Spec Tip
Luer
Sub Group
Standard
Spec Gauge
23G
Units per box
50/box
BC Butterfly 23G 3/4"
BC2334B
Spec length
-
Spec length
3/4"
Spec Tip
Luer
Sub Group
Standard
Spec Gauge
23G
Units per box
100/box
Safety Pen-type Blood Needle 22G 1 1/2"
BC22112PS
Spec length
-
Spec length
1 1/2"
Spec Tip
Safety Pen
Sub Group
Standard
Spec Gauge
22G
Units per box
48/box
Huber Needle 22G 25mm
HBN2225
Spec length
-
Spec length
25mm
Spec Tip
-
Sub Group
Huber
Spec Gauge
22G
Units per box
50/box
Huber Needle 22G 20mm
HBN2220
Spec length
-
Spec length
20mm
Spec Tip
-
Sub Group
Huber
Spec Gauge
22G
Units per box
50/box
Safety Pen-type Blood Needle 22G 1"
BC2201PS
-
Spec length
1"
Spec Tip or Cone
Safety Pen
Sub Group
Standard
Spec Gauge
22G
Units per box
48/box
Pen-type Blood Needle 22G 1 1/2"
BC22112P
-
Spec length
1 1/2"
Spec Tip or Cone
Pen Type
Sub Group
Standard
Spec Gauge
22G
Units per box
100/box
Pen-type Blood Needle 22G 1"
BC2201P
-
Spec length
1"
Spec Tip or Cone
Pen Type
Sub Group
Standard
Spec Gauge
22G
Units per box
100/box
Safety Butterfly Needle 21G 3/4"
BTF2134S
-
Spec length
3/4"
Spec Tip or Cone
Luer
Sub Group
Standard
Spec Gauge
21G
Units per box
100/box
Safety Needle 21G 1"
DN2101S
-
Spec length
1"
Spec Tip or Cone
Luer
Sub Group
Safety
Spec Gauge
21G
Units per box
100/box
Safety Needle 21G 1 1/2"
DN21112S
-
Spec length
1 1/2"
Spec Tip or Cone
Luer
Sub Group
Safety
Spec Gauge
21G
Units per box
100/box
Safety Needle 21G 1 1/4"
DN21114S
-
Spec length
1 1/4"
Spec Tip or Cone
Luer
Sub Group
Safety
Spec Gauge
21G
Units per box
100/box
Disposable Needle 21G 1 1/2"
DN21112
-
Spec length
1 1/2"
Spec Tip or Cone
Luer
Sub Group
Standard
Spec Gauge
21G
Units per box
100/box
Disposable Needle 21G 1"
DN2101
-
Spec length
1"
Spec Tip or Cone
Luer
Sub Group
Standard
Spec Gauge
21G
Units per box
100/box
Butterfly Needle 21G 3/4"
BTF2134
-
Spec length
3/4"
Spec Tip or Cone
Luer
Sub Group
Standard
Spec Gauge
21G
Units per box
100/box
Safety Pen-type Blood Needle 21G 1 1/2"
BC21112PS
-
Spec length
1 1/2"
Spec Tip or Cone
Safety Pen
Sub Group
Standard
Spec Gauge
21G
Units per box
48/box
Safety Pen-type Blood Needle 21G 1"
BC2101PS
-
Spec length
1"
Spec Tip or Cone
Safety Pen
Sub Group
Standard
Spec Gauge
21G
Units per box
48/box
Safety BC Butterfly 21G 3/4"
BC2134BS
-
Spec length
3/4"
Spec Tip or Cone
Luer
Sub Group
Standard
Spec Gauge
21G
Units per box
50/box
Pen-type Blood Needle 21G 1 1/2"
BC21112P
-
Spec length
1 1/2"
Spec Tip or Cone
Pen Type
Sub Group
Standard
Spec Gauge
21G
Units per box
100/box
Pen-type Blood Needle 21G 1"
BC2101P
-
Spec length
1"
Spec Tip or Cone
Pen Type
Sub Group
Standard
Spec Gauge
21G
Units per box
100/box
BC Butterfly 21G 3/4"
BC2134B
-
Spec length
3/4"
Spec Tip or Cone
Luer
Sub Group
Standard
Spec Gauge
21G
Units per box
100/box
Safety Needle 20G 1 1/2"
DN20112S
-
Spec length
1 1/2"
Spec Tip or Cone
Luer
Sub Group
Safety
Spec Gauge
20G
Units per box
100/box
Huber Needle 20G 20mm
HBN2020
-
Spec length
20mm
Spec Tip or Cone
-
Sub Group
Huber
Spec Gauge
20G
Units per box
50/box
Huber Needle 20G 40mm
HBN2040
-
Spec length
40mm
Spec Tip or Cone
-
Sub Group
Huber
Spec Gauge
20G
Units per box
50/box
Safety Needle 19G 1"
DN1901S
-
Spec length
1"
Spec Tip or Cone
Luer
Sub Group
Safety
Spec Gauge
19G
Units per box
100/box
BC Butterfly 20G 3/4"
BC2034B
-
Spec length
3/4"
Spec Tip or Cone
Luer
Sub Group
Standard
Spec Gauge
20G
Units per box
100/box
Safety Needle 19G 1 1/2"
DN19112S
-
Spec length
1 1/2"
Spec Tip or Cone
Luer
Sub Group
Safety
Spec Gauge
19G
Units per box
100/box
Huber Needle 19G 20mm
HBN1920
-
Spec length
20mm
Spec Tip or Cone
-
Sub Group
Huber
Spec Gauge
19G
Units per box
50/box
Disposable Needle 19G 1 1/2"
DN19112
-
Spec length
1 1/2"
Spec Tip or Cone
Luer
Sub Group
Standard
Spec Gauge
19G
Units per box
100/box
Safety Needle 18G 1"
DN1801S
-
Spec length
1"
Spec Tip or Cone
Luer
Sub Group
Safety
Spec Gauge
18G
Units per box
100/box
Disposable Needle 19G 1"
DN1901
-
Spec length
1"
Spec Tip or Cone
Luer
Sub Group
Standard
Spec Gauge
19G
Units per box
100/box
Safety Needle 18G 1 1/2"
DN18112S
-
Spec length
1 1/2"
Spec Tip or Cone
Luer
Sub Group
Safety
Spec Gauge
18G
Units per box
100/box
Blunt Needle 18G 1" (Filter)
BLN1801F
-
Spec length
1"
Spec Tip or Cone
Luer
Sub Group
Standard
Spec Gauge
18G
Units per box
100/box
Blunt Needle 18G 1 1/2" (Filter)
BLN18112F
-
Spec length
1 1/2"
Spec Tip or Cone
Luer
Sub Group
Standard
Spec Gauge
18G
Units per box
100/box
Blunt Needle 16G 1"
BLN1601
-
Spec length
1"
Spec Tip or Cone
Luer
Sub Group
Standard
Spec Gauge
16G
Units per box
100/box
Safety Pen-type Blood Needle 18G 1 1/2"
BC18112PS
-
Spec length
1 1/2"
Spec Tip or Cone
Safety Pen
Sub Group
Standard
Spec Gauge
18G
Units per box
48/box
Blunt Needle 16G 1 1/2"
BLN16112
-
Spec length
1 1/2"
Spec Tip or Cone
Luer
Sub Group
Standard
Spec Gauge
16G
Units per box
100/box
Pen-type Blood Needle 18G 1 1/2"
BC18112P
-
Spec length
1 1/2"
Spec Tip or Cone
Pen Type
Sub Group
Standard
Spec Gauge
18G
Units per box
100/box
Blunt Needle 18G 1"
BLN1801
-
Spec length
1"
Spec Tip or Cone
Luer
Sub Group
Standard
Spec Gauge
18G
Units per box
100/box
Blunt Needle 18G 1 1/2"
BLN18112
-
Spec length
1 1/2"
Spec Tip or Cone
Luer
Sub Group
Standard
Spec Gauge
18G
Units per box
100/box